摘要
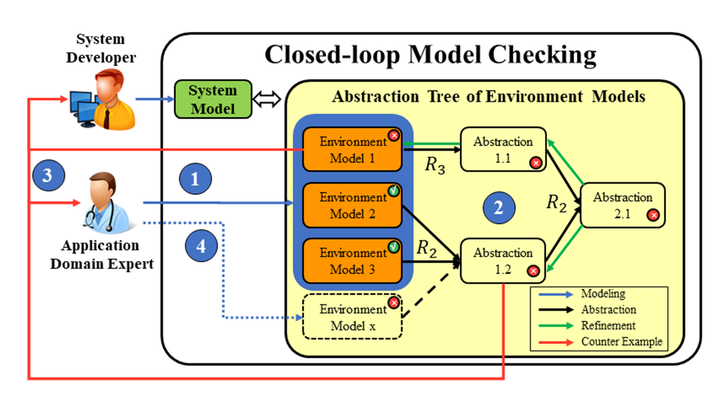
Bringing new safety-critical medical devices to market faces several major challenges, but modeling and formal methods can facilitate this process from early system requirements verification to platform-level testing to late-stage clinical trials.
类型
出版物
Computer